A story that starts out claiming that alternative phenotypes are excellent to study eco-evolutionary feedbacks, but ditches this idea to end on the notion that eco-evolutionary change is centred around evolving developmental systems. It is a personal journey of stepping out of the comfort zone of mainstream eco-evolutionary thinking, and of formulating arguments to convince myself, and hopefully others, to explore new concepts to study eco-evolutionary dynamics.
* Smallegange, I.M. (2022). Integrating developmental plasticity into eco-evolutionary population dynamics. Trends in Ecology & Evolution 37:129-137
2017-2018: Alternative phenotypes are excellent systems to study eco-evolutionary feedback loops!
Most studies on eco-evolutionary dynamics have focused on interactions between predators and their prey, or between parasites and their hosts (Govaert et al. 2019). Less is known about how eco-evolutionary interactions affect trait dynamics within single populations. Within single populations, both ecological and evolutionary variables can be both the drivers and the objects of change. This means that we can distinguish an ecology-to-evolution pathway and an evolution-to-ecology pathway that together comprise an eco-evolutionary feedback loop (Figure 1):

Daphne Vink and Tim van Gorcum, two Master students, and I reviewed the literature and found that eco-evolutionary feedback loops have been identified for alternative phenotypes which expression depends strongly on how genes interact with population density. Examples include behaviour driven by a cooperative gene in budding yeast (Sanchez & Gore 2013), the genetically determined mode of reproduction (sexual or asexual) of the rotifer Brachionus calyciflorus (Stelzter 2012), genetic clones of the green peach aphid (Myzus persicae) that differ in reproduction and survival rates (Turcotte et al. 2013), and genetically determined throat colours of female side blotched lizards (Uta stansburiana) that are associated with different reproductive strategies (Sinervo et al. 2000); all of which vary in frequency with varying population density, and vice versa. This all seemed very promising!
2019: Alternative phenotypes are not at all suited to study eco-evolutionary feedback loops!
However, in addition to genetic influences, expression of most alternative phenotypes depends strongly on environmental cues, including population density (Smallegange et al. 2019), that affect the development of phenotypes. So what does that mean for the premise that alternative phenotypes are excellent systems to study eco-evolutionary feedback loops? It introduces an extra level of complexity: the developmentally plastic expression of phenotypes depends on the environment (in this case population density), and vice versa, influencing the eco-evolutionary feedback loop (Figure 2). It means we need to integrate developmental plasticity into the eco-evolutionary framework. But how?
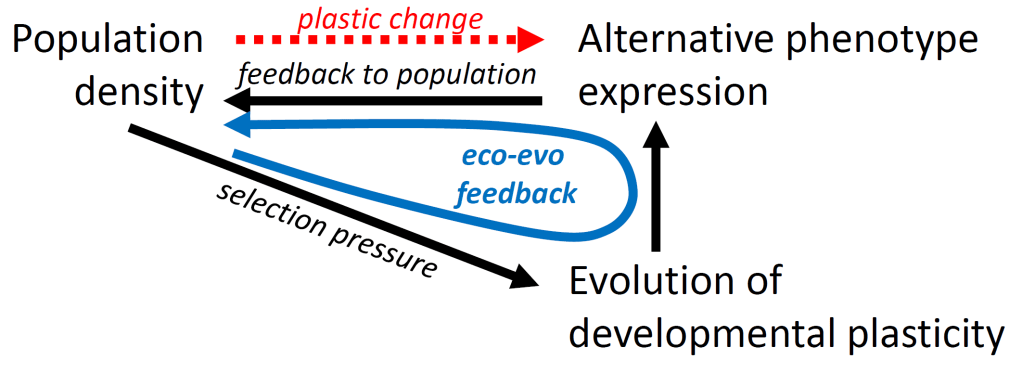
One solution, put forward by a colleague, is to not attempt this at all, because eco-evolutionary dynamics is defined as the study of how genetically determined phenotypes influence ecological variables, and vice versa (Figure 1; Hendry 2017), excluding (strong) plasticity. But I find this far from satisfactory, and I am not the only one advocating we should include plasticity into the eco-evolutionary framework (Hendry 2017). Plasticity is central to eco-evolutionary dynamics because population dynamics emerge from birth and death rates that are determined by phenotypes in interaction with their environment.
Another solution is to use genotype-by-environment interactions that describe how the target phenotype for a specific genotype varies as a function of the developmental environment. This is a good and practical solution if phenotype expression in your study system is (strongly) gene-based. But it did not help me to understand the eco-evolutionary dynamics of the bulb mite populations in my lab (Smallegange et al. 2019). I wanted to focus on the actual developmental process, instead of the outcomes of development.
2020: COVID-19 brings challenges but also opportunities
The manuscript that advocated some of the above ideas was firmly rejected by reviewers and editor: our arguments were completely incomprehensible, even after revision. Ouch. The whole thing needed a rethink.
Then, COVID-19 arrived, putting a halt on revising the manuscript. Home schooling my son and student teaching took priority. Fortunately, I was able to continue literature discussions with my small group of PhD and Master students online. In particular Flor Rhebergen was instrumental in providing food for thought. We started on a book “Cycle of Contingency: Developmental Systems and Evolution” by Oyama et al. (2001). The book introduced us to developmental systems theory and a perspective that is centred around the fundamental role of development in the realisation of phenotypes.
More books followed: Evolutionary Causation (Uller & Laland 2019), Organism & Environment: Ecological development, Niche Construction and Adaptation (Sultan 2017), Ecological Developmental Biology: Integrating Epigenetics, Medicine, and Evolution (Gilbert & Epel 2009), Developmental Plasticity and Evolution (West-Eberhard 2003), among others.
The COVID-19 outbreak also meant that conferences and seminars were moving online. I typically do not travel to attend these because of care duties (see e.g. this post), but now I could virtually attend seminars by those more involved with developmental system perspectives: developmental psychologists and philosophers of science. It was all-inspiring!
2021: Paper accepted!
At some point I was directed towards a paper by Gilbert Gottlieb on probabilistic genetics (Gottlieb 2007), where he describes the developmental process as characterized by a reciprocity of influences within and between the levels of the developmental manifold of an individual (Figure 3).
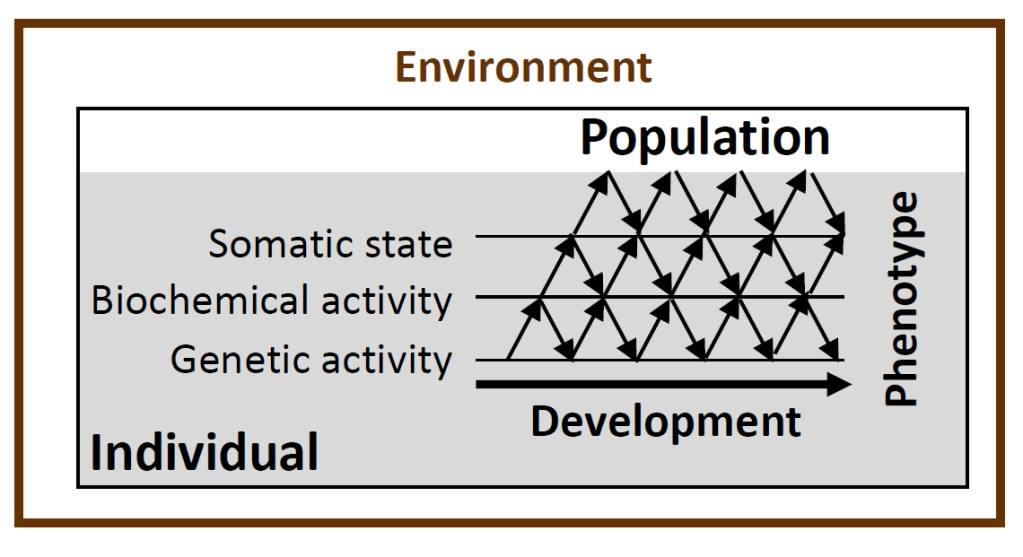
Based on Gottlieb (2007), I formulated some more, I hope, comprehensible notions on how to integrate developmental plasticity into eco-evolutionary dynamics. Hooray! I was eager to continue this process with a co-author, but those that I approached, politely declined as they were too busy, or found the subject matter too alien.
Undeterred, after some friendly fire from colleagues, I submitted the now single-author manuscript to Trends in Ecology and Evolution, where, this time round, it sailed through the review process. The future will tell how it lands.
2022: Epilogue
I look back on a very inspiring journey, where I met new colleagues, fostered new collaborations and had very insightful and lively discussions, exploring different literatures. It has culminated in ideas for a new research programme that I aim to start at my new workplace at Newcastle University – home to two scholars, Daniel Nettle and Melissa Bateson, whose paper “Adaptive developmental plasticity: what is it, how can we recognize it and when can it evolve?” (Nettle & Bateson 2015) sparked this whole adventure.
References
Gilbert, S. and Epel, D. (2009) Ecological Developmental Biology: Integrating Epigenetics, Medicine, and Evolution, Sinauer Associates.
Gottlieb, G. (2007) Probabilistic genetics. Dev. Sci. 10, 1–11.
Govaert, L. et al. (2019) Eco-evolutionary feedbacks – theoretical models and perspectives. Funct. Ecol. 33, 13–30
Hendry, A.P. (2017) Eco-Evolutionary Dynamics, Princeton University Press.
Kokko, H., & López‐Sepulcre, A. (2007). The ecogenetic link between demography and evolution: can we bridge the gap between theory and data? Ecology Letters, 10, 773–782.
Nettle D, Bateson M. (2015). Adaptive developmental plasticity: what is it, how can we recognize it and when can it evolve? Proc. R. Soc. B 282: 20151005.
Oyama, S. et al., eds (2001) Cycle of Contingency: Developmental Systems and Evolution, MIT Press.
Sanchez A., & Gore, J. (2013). Feedback between population and evolutionary dynamics determines the fate of social microbial populations. PLoS Biology, 11, e1001547.
Sinervo, B., Svensson, E., & Comendant, T. (2000). Density cycles and an offspring quantity and quality game driven by natural selection. Nature, 406, 985–988.
Stelzer, C.P. (2012). Population regulation in sexual and asexual rotifers: an eco‐evolutionary feedback to population size? Functional ecology, 26, 180–188.
Smallegange, I.M. et al. (2019) Cross-level considerations for explaining selection pressures and the maintenance of genetic variation in condition-dependent male morphs. Curr. Opin. Insect Sci. 36, 66–73
Sultan, S.E. (2015) Organism & Environment – Ecological Development, Niche Construction and Adaptation, Oxford University Press.
Turcotte, M. M., Reznick, D. N., & Hare, J. D. (2013). Experimental test of an eco-evolutionary dynamic feedback loop between evolution and population density in the green peach aphid. The American Naturalist, 181, S46–S57.
Uller, T., Laland, K.N., eds (2019) Evolutionary Causation: Biological and Philosophical Reflections, MIT Press.
West-Eberhard, M.J. (2003) Developmental Plasticity and Evolution, Oxford University Press.
One Comment Add yours