We are in great need of an integrative framework that allows ecologists to predict life history strategies (i.e. the different ways in which individuals trade-off resource investment into survival or reproduction) from functional traits: traits of individuals that inform on the performance of a population of plants or animals as a whole. To contribute towards building such a framework, the scientific journals Journal of Animal Ecology, Journal of Ecology and Functional Ecology, all from the British Ecological Society (BES), have just published a cross-journal special feature entitled “Linking organismal functions, life history strategies and population performance”, edited by Roberto Salguero-Gómez, Cyrille Violle, Olivier Gimenez, and Dylan Childs.
Why does this special feature focus on using traits of individuals to predict what happens to populations and species in the face of changes in the environment? Firstly, one of the traits of individuals that is very often scored in field and lab studies is a measure of body size, like body length. So, it would be highly efficient if we could predict the fate of populations from such an easy-to-measure quantity. Secondly, environmental effects are often mediated through the size of individuals. For example, if food availability declines, individuals of a population will lose mass and eventually die if resource availability reaches critically low levels. Furthermore, shifts in body size at the population-level can carry over to affect larger scale dynamics. E.g., changes in mean body size of species in food webs are known to alter the resilience of the system to disturbance [1], as well as altering the strength of trophic cascades [2]. This is why body size is often considered a functional trait. Finally, including information on body size dynamics into traditional indicators of population decline and extinction can produce more accurate inferences of whether a population is approaching such a critical transition than using traditional indicators based on abundance time-series alone [3].
In one of the papers of this special feature, I, together with my then master student Hedwig Ens, investigated if my recently developed population model [4] can be used to accurately predict the structure and size of populations of bulb mites (Rhizoglyphus robini) after selectively harvesting large individuals from single populations [5]. The main drivers of the growth or decline of a population are the demographic rates: the rates with which individuals survive, grow and reproduce over a certain time period, for example a year. These demographic rates, in turn, depend on the traits of individuals, for example their body size. Thus, this type of model is referred to as a trait-based population model. The novelty of this recently developed population model [4] is that, contrary to its predecessors, the demographic rates of growth and reproduction are determined by a growth model [6] that takes into account the fact that individual animals can spend energy only once: if an individual has used energy from ingested food for growth, it cannot be used for reproduction, and vice versa. Crucially, this growth model is a model that predicts the growth and reproduction of individuals. The big question is whether a model that describes the growth and reproduction of individuals well, also accurately describes the performance of a population of individuals. Jaap van der Meer, for example, has recently shown in a theoretical paper [7] that the assumptions that are made to make the growth model work well at the individual level, result in a population model that overestimates the abundance of juveniles, but underestimates the abundance of adults. This is the opposite of what we generally observe in nature where animal populations are dominated by adults instead of juveniles.
| Figure 1. Our experimental bulb mite populations are kept in small tubes and can consist of hundreds of individuals, depending on food conditions. |
So, back to our paper, where Hedwig and I set out to test the performance of our population model. We parameterised the model for our favourite model species, the bulb mite (Rhizoglyphus robini), and created predictions on the size and structure (proportion of eggs, juveniles and adults) of the mite populations under different types of change in the environment. We then compared these predictions with observations on our experimental bulb mite populations (Figure 1).
We found that the model very well predicted how total population size varied over time (Figure 2). We also found that variation in population size, i.e. whether it would increase or decrease, depended greatly on the proportion of days in the experiment that the mite populations received food: the higher this proportion, the higher the population counts. However, like Jaap van der Meer already found [7], the model predicted a relatively higher abundance of juveniles than adults.
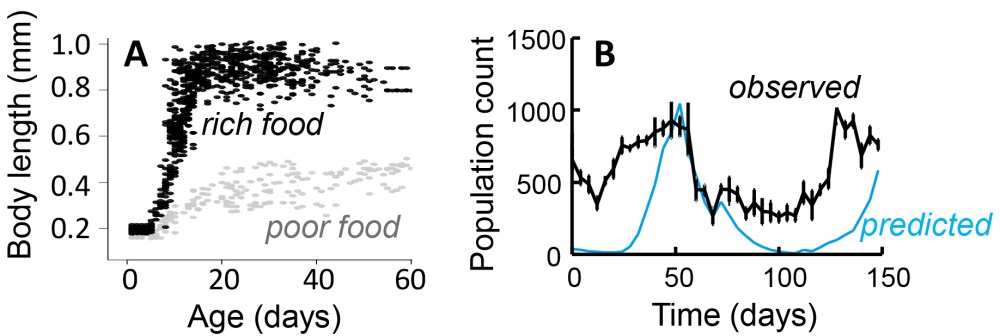
| Figure 2. Food quality greatly affects R. robini growth curves [8] (A), but we are able to accurately predict how populations change over time in response to variation in food [5] (B) |
So where does that leave us? For now I am satisfied to use to model to predict overall population performance: under what circumstances is a population predicted to decline, and when to grow. However, there is still the problem that the model does not accurately predict the structure of populations as the proportion of juveniles is overestimated and the proportion of adults underestimated. I have recently submitted a grant proposal to the European Research Council that was aimed at tackling this problem, but this was rejected last week. Solving this mismatch between a well-performing growth model of individuals that does not predict the structure of populations well [5, 7], will have to wait for now. Hopefully, though, this cross-BES journal special feature will emphasize the need for developing models that we can use to predict both population structure as well as size.
References
1 Woodward G, Ebenman B, Emmerson M, Montoya JM, Olesen JM, Valido A, Warren, PH. 2005. Body-size in ecological networks. Trends in Ecology and Evolution 20: 402–409.
2 DeLong JP, Gilbert B, Shurin JB, Savage VM, Barton BT, Clements CF, Dell AI, Greig HS, Harley CD, Kratina P, McCann KS, Tunney TD, Vasseur DA, O’Connor MI. 2015. The body size dependence of trophic cascades. American Naturalist 185:354-66
3 Clements C, Ozgul A. 2016. Including trait-based early warning signals helps predict population collapse. Nature Communications 7: 10984.
4 Smallegange IM, Caswell H, Toorians MEM, de Roos AM. 2017. Mechanistic description of population dynamics using dynamic energy budget theory incorporated into integral projection models. Methods in Ecology and Evolution 8: 146-154.
5 Smallegange IM, Ens HM. 2018. Trait-based predictions and responses from laboratory mite populations to harvesting in stochastic environments. Journal of Animal Ecology doi:10.1111/1365-2656.12802
6 Kooijman, SALM, Metz JAJ. 1984. On the dynamics of chemically stressed populations: the deduction of population consequences from effects on individuals. Ecotoxicology and Environmental Safety 8: 254–274.
7 Van der Meer, J. 2016. A paradox in individual-based models of populations. Conservation Physiology 4: Issue 1.
8 Smallegange IM. 2011. Complex environmental effects on the expression of alternative reproductive phenotypes in the bulb mite. Evolutionary Ecology 25: 857-873.